For complete guidelines and resources for transporting COVID-19 test samples, please visit our coronavirus shipping guidelines page.
With new COVID-19 test sites setting up all over the United States and the world, many businesses and their employees will be transporting infectious substances for the first time.
These shippers must be aware of two important topics:
- In general: how to ensure their shipments comply with Dangerous Goods transport regulations
- Specifically: how to ensure their test samples are not rejected by testing laboratories
Two Labelmaster experts—Manager of Regulatory Affairs and Corporate Responsibility Brian Beetz and Senior Manager Jay Johnson—recently shared their insights and knowledge about these critical topics.
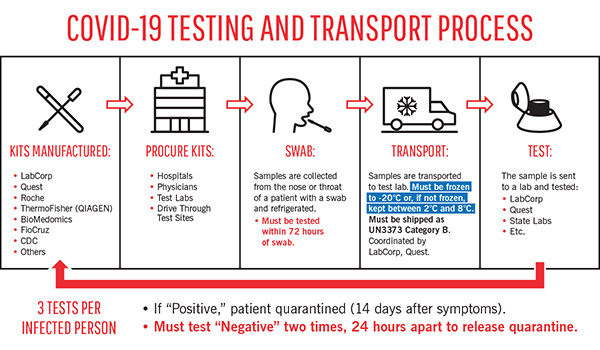
COVID-19 test samples are Biological Substances, Category B (UN 3373)
Infectious substances are classified as Dangerous Goods, which makes them subject to domestic and international shipping regulations for packaging and labeling for hazardous materials.
The good news is that COVID-19 test samples are not difficult to ship compliantly. (Though please see the update below.) “COVID-19 test samples are classified in the U.S. as Biological Substance, Category B” says Beetz, “which means they are not generally capable of causing permanent disability or life-threatening or fatal disease in otherwise healthy humans. Category B biological substances are relatively easy to transport in a compliant manner.”
(Category A infectious substances such as Ebola, which are considered immediately life-threatening, are governed by far more restrictive regulations.)
UPDATE: The World Health Organization (WHO) has assigned COVID-19 or SARS-CoV-2 virus clinical (non-propagated) samples to Category B and cultures/isolates to Category A while the United States has assigned both (i.e. clinical and cultures) samples to Category B.
This means that as long as the intended shipment of cultures is within the U.S., you can send it as Cat B. But if either the origin, destination or transit country is issuing a different policy, then you must observe it.
The Centers for Disease Control and Prevention offers this guideline for COVID-19 specimen packing and shipping:
Pack and ship suspected and confirmed SARS-CoV-2 patient specimens as UN 3373 Biological Substance, Category B, in accordance with the current edition of the International Air Transport Association (IATA) Dangerous Goods Regulations. Personnel must be trained to pack and ship according to the regulations and in a manner that corresponds to their function-specific responsibilities.
(emphasis ours)
“All the Category B kits we offer for shipping COVID-19 test samples include the UN 3373 mark,” says Beetz. He also points out that if your samples are packed with dry ice, that dry ice itself is regulated hazmat by air—Class 9, UN 1845, to be exact.
How Category B makes shipping COVID-19 test samples easier
Johnson notes that Category B biological substance transport can be much less complicated than many other hazmat shipments. “You don’t have to create a Shippers Declaration for Dangerous Goods (DGD), and there’s no hazmat surcharge. You don’t need UN-specification packaging, which is generally more expensive. Also, dry ice is only regulated if you’re shipping it by air since solid carbon dioxide sublimating displaces oxygen.”
Regarding patient sample collection, Johnson says “there is an exception in the 49 CFR for Category B patient samples being transported exclusively by private or contract carriers. But if you use standard carriers like FedEx and UPS you will have to comply with all requirements, including training. Category B training courses are available online and in custom webinar format.”
A difficult temperature range for COVID-19 test samples
According to the CDC, COVID-19 test samples must be kept frozen or between 2 – 8 °C (35.6 – 46.4 °F) at all times. Samples that do not stay in this range will be rejected, since they are more likely to give a “false negative” result. (This is why it’s essential to include a Cold Chain Complete Temperature Indicator for shipping each sample.)
Johnson explains why maintaining these temperatures can prove difficult. “Think about why you put ice cubes in a drink—as the ice melts (changes phase) the energy is taken from the liquid around the ice lowering the temperature of the drink toward the phase change temperature of 0 °C. In temperature-controlled packaging you’re more likely to have deviation at the low end. Too much frozen water—ice—changing phase at 0 °C can cool your biological samples below the 2°C limit of your required temperature range.
“There’s no perfect material that changes phase at, say, 4°C , so your samples need to be buffered with chilled gel bricks. You can use a variety of insulated packaging, and the better the material, the less coolant you need.
“The most important thing is to test your package against the expected transport temperature range, and then adjust your phase change material until your sample stays in the required range.”
For complete guidelines and resources for transporting COVID-19 test samples, please visit our coronavirus shipping guidelines page.Questions? Please call us at 800.621.5808.
Make sure your shipments are safe and in complete compliance with a full line of solutions from Labelmaster—a full-service provider of goods and services for hazardous materials and Dangerous Goods professionals, shippers, transport operators and EH&S providers.


